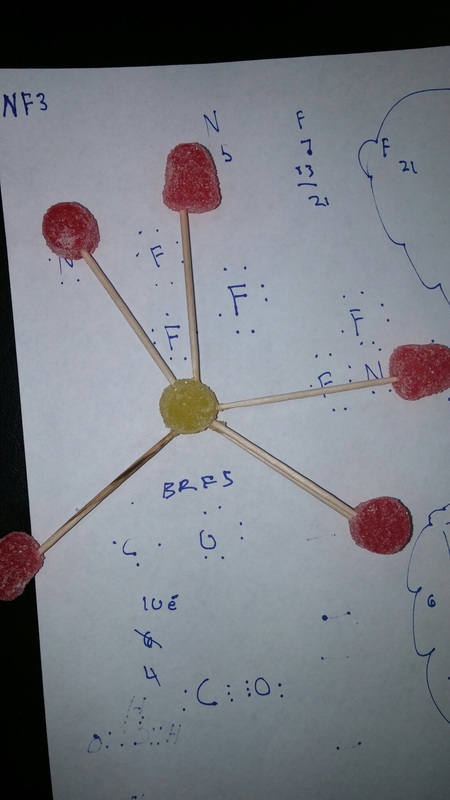
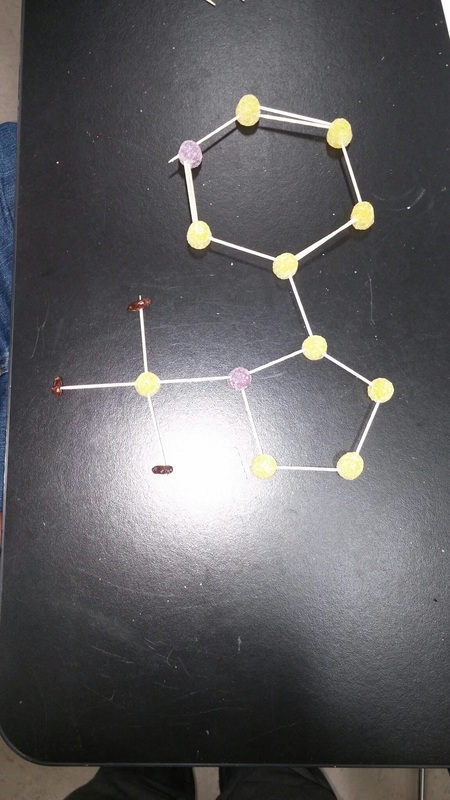
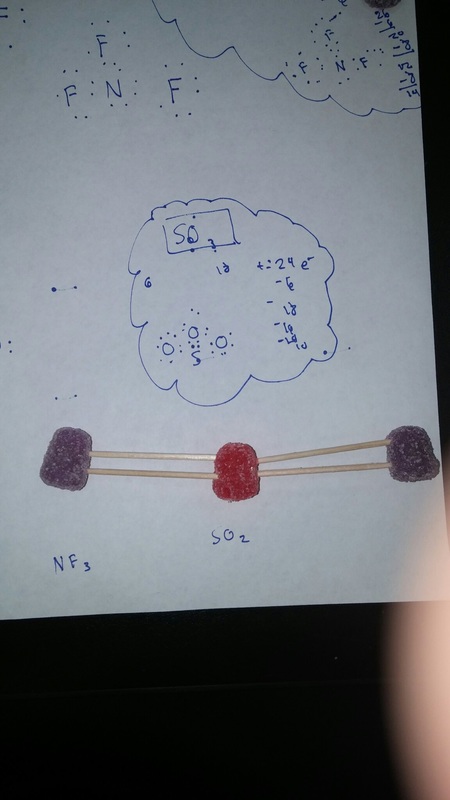
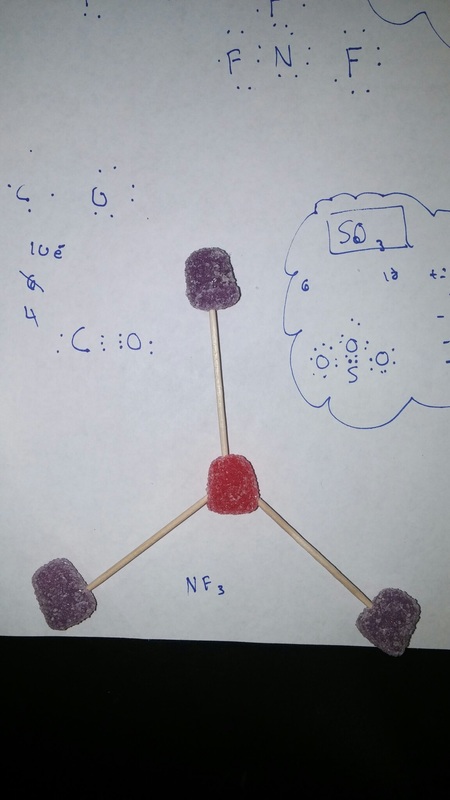
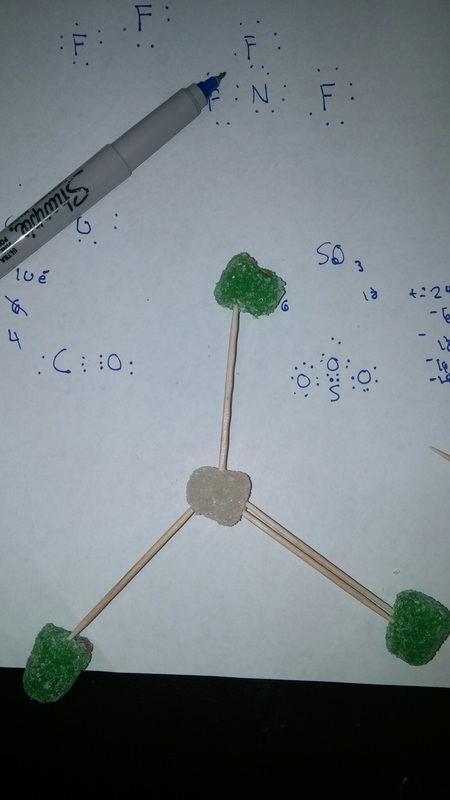
For the the gumdrop lab we looked organic and inorganic molecules. Our objective was to get a hands on experience and to get a better understanding on how they are made. We used gumdrops as atoms, raisins as hydrogen, and toothpicks as the bonds. With these materials we created the compounds. Through this experiment I got a better understanding on how the compounds are formed. With the hands on learning I felt like I got a closer on how the compounds are formed. Some things that learned about that compounds was that carbon form rings that look like hexagons on there molecular structure. They make this shape because carbon is stable. A conclusion that I can draw from the end of this experiment is that molecules take similar shapes to stay as far away from each other.
In the beginning of this project I really wasn't that interested in it. My friend asked me to do it with him and saw other people have fun, so I kept an open mind and tried it. When picking the rocks I really had no idea what I wanted, my friend told me that labradorite would like nice so I picked up a piece. Prior to cutting our rocks my friends and I researched the rock. We looked up it's characteristics, what makes it unique, and it's cleavage. We found out that with labradorite if your are lucky that when the light reflects on the rock it gives you multiple reflecting colors. Once finished researching I couldn't wait to start. When I started out with my rock I didn't think much of it I didn't think that it would be all that special. While waiting the cut my rock Andrew offered to cut mine for me so I accepted. I didn't know how to do it so I thought I would learn from a professional. Once it was finished, it was like opening gifts on Christmas. The rock looked amazing, it was vibrant and full of transparent colors. Andrew told me it was the best looking piece, so now my mission was to make it the best looking rock possible. At first I was doing a pretty good job sanding it. Minutes later I amazingly failed at my job. The rock slipped and stuck in the sander. There was a huge scratch down the middle of it, It was like dropping the winning touchdown pass. I felt terrible but I knew I had to make it better. So after sanding it for a couple of days I finally got the finished piece. It was amazing and I was proud to say that I made that. I then decided to give it to my Mom because I knew she would love it. Overall it was a fun experience. Looking Back: Looking back at the experiment I got good results. In the beginning I ran through some bumps in the road when trying to make it perfect but I overcame those obstacles. I thought the finished look of the rock looked amazing, I was more than satisfied with the results I got. Looking Beyond: When coming to the science aspect of the rock it's really interesting to me. The way labrodorite works is that the light reflects on different twinnings of the rock . The color that shows depends on inner twinning, if your are lucky you can get a multi color look to it. In a nut shell depending what's on the inside determines what colors would be reflective in the rock. Looking Inward: Overall in this experiment I got desirable results. The rock looked great and I couldn't be happier with it. It's full of color and shines beautifully. Overall I'm proud of the results that I got. looking Outward: When I did this project I did it on my own with some help by the people around me. The part that took group collaboration was actually sharing the lapidary machine. Their was instances where their was conflict, but I believe that I handled it well. I'm a lover not a fighter so I'm not one to get aggressive when it comes to sharing things so I tried to facilitate the machine well. I made sure that every one had a chance and that there was only three people. In general it went well for me. Looking Forward: If I could do this again there wouldn't me much that I would change. I mean not to brag or anything but I believe the rock looks amazing. But the one things that I would go back and do would to polish it a bit more. I think it could use a bit more, but I don't think it takes much from the overall beauty of it. Take Aways: Some take aways that I got from this experiment is the variety is the spice of life. Never had I thought of polishing and shaping rocks, but after this project it would be something that I would like to continue doing. It was a fun project and I had a great time doing it. So far this semester was a blast. I had so much fun in chemistry and I learned a lot about my self as a learner. I went through some struggles early on with the grading system but in the end I believe that I adapted well to the system. Overall this semester was hands down one of my favorites.
This semester I learned about myself. At first I was a but worried about the grading system because it was new to me. It was really the first class where I was in control of what I was learning. I felt that when I walked into that class that it was my time in their. I learned what I wanted to learn and if I waisted time I was wasting my time. At first I was a little stressed about that because I really never had a class where I was in control of my learning. After a couple of weeks I really got the hang of it. Coming into this class I knew I had time management skills but I lacked the confidence of believing in myself. But looking back I gained the confidence within in my self and I believe that I can take on the world. I also kind of got a feel of what it would be like in college. No one it's going to tell you to do your own work, you're in charge of that and I get like I honed in those skills. In the end I gained confidence within my self. This Semester I really felt that I was motivated by my curiosity. I really ventured into my own self interests and did experiments that I thought were interesting to me. Occasionally I did worksheets when I really had to but I tried to avoid them when possible. For example I saw a Rubens tube and in a snap it caught my attention. Another example was the rock project. I heard and saw people doing it and it seemed interesting to me so I tried it. The end result was amazing I got this one of kind rock out of it and inspired me to appreciate beauty of what could be made from rocks. In this class I will take away many things. This class really spiked my curiosity with science. Never have I wondered about the stars out there or how rockets lift off the ground. I also learned the importance of how you affect your learning. What you put into a class is what you will get out of it. For me I tried many different things and with that I had an amazing experience in chemistry class. Honestly this is one of the only classes where I was excited to come into everyday. In conclusion I learned a lot about my self and just had fun. Today in class we finished our three day mission on finishing our Rubens tube. It took a lot of hard work and labor but we finally completed it. Some background knowledge is that a Rubens' tube is a standing wave flame tube, or simply flame tube. It is obsolete in today's standards of physics apparatus for demonstrating acoustic standing waves in a tube. Never the less it's still fun to look at. It was invented by German physicist Heinrich Rubens in 1905, it graphically shows the relationship between sound waves and sound pressure, like an oscilloscope. We decided to do this after watching countless videos on YouTube and we then decided to take matters into our own hands. The first couple of days just consisted of waiting for parts to arrive and learning about how it worked. The remainder of the time we worked as fast as we could to try to get it done. Overall this was my favorite project by far. This is work I am proud to show off and be enthusiastic about.
Looking Back: Overall I'd say we got really desirable results. At first the tube was working when we put music in it but it was a little sketchy. It worked but it kept running out of fire. So our next step was then to add another propane tank. After adding the propane we got the results we were looking for. It was amazing you could see the sound waves moving through the tube as the music was playing. We even got to see two peaks emerge as music was playing. Overall after adding an additional propane tank we were able to get the results we were looking for. Looking Beyond: The basic principles on how this experiment worked was by sound waves and vibrations. As the sound vibrations it would cause pressure to vary within the tube causing the fire to go up and down. Using Bernoulli's principle, the gas flow is proportional to the square root of the pressure difference between the inside and outside of the tube. With this being said the flame height depends non-linearly on the local, time-dependent pressure. The time average of the flow is reduced at the points with oscillating pressure and thus flames are lower. So in conclusion more pressure equals more flame height. Looking Inward: I strongly believe that my group got desirable results. Everything might have no worked out perfectly at first but after a couple of trials we were able to achieve what we wanted. In this experiment I believe that I performed really well. I worked hard on it every day and never gave up on it. Their might of been times were all hope seemed lost but I kept telling myself that we would get it done in the end. Looking Outward: In this project I I worked with Bryan, Jake, and Josh. I would have to say this is the best group that I have been in. We were all determined to get the tube working. We also split the work up and made sure that everyone had something to do while working on the project. Our group stayed together the whole time and we didn't run into any issues with each other. Overall we collaborated well and we had a fun time doing so. Looking Forward: When thinking about what could been better nothing really comes into mind. I mean we dealt with struggles but we overcame them and everything worked as planned. With that being said I would like to take this to the next step. There was this video on YouTube where they took the tube to the next level and made a 3D box that still did the same thing as a Ruben tube but better. It's something that I would like to try maybe in the future. Take Aways: Some take a ways that I got from this experiment was to never give up. Sure it seems like a cliche thing to say but it actually worked out for us. We just kept grinding away and never gave up. In the end it actually worked and this is a project that I will never forget. It was so much fun making it and then end result was priceless. Overall the take away I got was to have fun and never give up when all hope seemed lost. link https://www.youtube.com/watch?v=ea90dHuibmk Today in class we started to actually launch rockets for the the first time. Previously we created rocket engines but we didn't get to actually launch them. This time using simple materials we had our chance to do it for the first time. But as always before we started the experiment we had to go through the key concepts of the lesson. We started of by going over Chemical Reactions. A chemical reaction is the process in which one or more substances changes into one or more substances. Also chemical reactions energy, and energy is either absorbed (endothermic) or given off (Exothermic). In addition we learned about the law of conservation. It states that for any system closed to all transfers of matter and energy, the mass of the system must remain constant over time, as system mass cannot change quantity if it is not added or removed. But what gets the rocket to launch is a reaction called a combustion reaction.In a combustion reaction compound and an oxidant is reacted to produce heat and a new product.
Today in class we started to actually launch rockets for the the first time. Previously we created rocket engines but we didn't get to actually launch them. This time using simple materials we had our chance to do it for the first time. But as always before we started the experiment we had to go through the key concepts of the lesson. We started of by going over Chemical Reactions. A chemical reaction is the process in which one or more substances changes into one or more substances. Also chemical reactions energy, and energy is either absorbed (endothermic) or given off (Exothermic). In addition we learned about the law of conservation. It states that for any system closed to all transfers of matter and energy, the mass of the system must remain constant over time, as system mass cannot change quantity if it is not added or removed. But what gets the rocket to launch is a reaction called a combustion reaction.In a combustion reaction compound and an oxidant is reacted to produce heat and a new product. Once the reaction occurs thrust makes the rocket lift off the floor, Thrust is sudden or violent lunge with a pointed tip. The point of this experiment was to learn more about combustion reactions and to to get us started with rockets. We used sugar and potassium nitrate for the engine, skewers for the body, a fuse to ignite it, and tape to put it all together. We started of by making the body of the rocket. We used paper and we rolled it up to make the body. We then tightened it up with tape to make sure that nothing slipped out. Our next step was to use tin foil to cap up the ends. Once the fuel was done being mixed we put the fuel inside the tubing and caped it up. Next we attached a fuse to the end and added a skewer to finish it up. Now the rocket was ready to launch. The point of the experiment was to learn more about combustion reactions and to see how they are used. Looking Back: Overall the experiment was pretty much a failure. I mean it was able to lift of the ground but with in seconds it came straight down. While building it I thought I would of gotten more out of it. I was really disappointed with my results but at the same time there was a bright spot towards it. The only bright spot that I got out of it was that I was able to at least get it off the ground. But in the end I did not get the results that I was hoping for. Looking Beyond: The science behind the experiment was really quite simple. The reaction that occurred was a combustion reaction and then some basics laws of gravity made it launch. In a combustion reaction compound and an oxidant is reacted to produce heat and a new product. Once the reaction occurs thrust makes the rocket lift off the floor, thrust is sudden or violent lunge with a pointed tip. Put all these together and you get a racket launch. Looking Inward: Since I worked alone I didn't really run into any group problems, but at the same time I wish I would of had someone there. It would of been nice to have someone to have some other input while building the rocket. Looking Forward: If I could do this lab again there would be a lot of different things that I wished I could changed. For example I would of made sure that I caped the ends better so that it could of had more thrust. I also would of made the rocket lighter. I noticed that I might have put to much tape on the rocket. I was just to nervous that maybe something might of come off, but in the end it might of hindered the overall performance of the rocket. Take Aways: Some take aways that I got from the experiment was learning more about rockets. It was a thrilling feeling waiting for the rocket to launch but the outcome if it made me disappointed. But it it only made me want it to try and again until I succeed. Overall it has inspired men to make more rockets. Today I watched a video of sorts science. I always enjoy watching the episodes because I like seeing how science is envolved with football. In the segmnet they talked about Calvin Johnson. I have always admired his ability to make spectacular catches based on his size. To start off they calculated that he is 2.5% slower to record of the 40 yard dash. He is 40 pounds heavier and it's significantly taller which makes the feat even more amazing. He also has a wingspan of 6ft 10in and also has a standing reach of 8ft 8in. Out all of his skills together he can cover 4,009 cubic feet, which is basically the size of a 2 car garage. Overall I like seeing how science can be used in the field of football.
Today in class we started to worked on making batteries. But before we started we went through some of the basic concepts. We learned that all elements have different attractions to elements, also known as electroneg activities. We also learned that when an atom that can lose electrons more easily than an atom of a different element that comes into contact with one another. One of the main concepts that we learned was about oxidation and reduction. It is a reaction in which there is a transfer of electrons. The difference between the two is that oxidation is the loss of electrons and reduction is the family of electrons. An example is when Cu and O come together, Copper becomes becomes oxidized and oxygen becomes reduced .
To start the battery we needed to use pennies, cardboard, and vinegar. Before we put anything together we had to make sure that one side of all the pennies was filed to where the zinc was visible. We then used cutouts for the pennies out of cardboard. We then dipped it into vinegar so that electrons would flow through from the zinc. Once completed we but the materials together. We then but the copper at the bottom, then zinc, and then cardboard for the electrolytes. We did this repeatedly so that we made use of all of our pennies. The repeated stacking increased the charge (volts) because they were arranged in series. The chemistry behind it that made the experiment work was that at contact with the electrolyte solution, each metal formed positive ions. Since zinc losses electrons more easily, they traveled through the copper ions. Looking Inward: Overall I feel like I got really desirable results. In the beginning we didn't get the results we wanted but after making revisions our results improved to where we wanted them to be. Looking Outward: I believe that I worked well with my group. We all worked together and we didn't run into problems. I believe that I contributed well and help complete the project. Looking Back: Everything did not work out as planned at the beginning and nothing worked. It was frustrating but we didn't give up hope. The next round we made sure we sanded the pennies down again and made cleaner cutouts. We also added more pennies so that we could get more volts for our battery. After the revisions it worked and the light was really bright. Looking Forward: If I could do this again something that I would do differently would be to add more pennies. The first time I added more pennies there was a significant difference and I would like to see more improvement. Looking Beyond: The chemistry behind it that made the experiment work was that at contact with the electrolyte solution, each metal formed positive ions. Since zinc losses electrons more easily, they traveled through the copper ions. Take Aways: Some take aways that got from the experiment was the importance of revisions. In the beginning of the experiment things weren't going the way as planned and it simply didn't work. After going back I made sure that the next draft everything was neat and the cut outs were more precise. Also making sure that the pennies were sanded down nicer. After the revisions it worked out better. The lesson learned after words was the importance of revisions. Unit 3:3 Ions- Take Home Quiz Questions
What is an ion? An Ion is an atom or molecule with a net electric charge due to the gain or loss of one or more electrons. What is a cation and an anion? A cation is a positive Ion that has lost one or more electrons. An anion is a negatively charged Ion that has more electrons than protons. Which elements typically form anions? Some elements that typically form anions are chloride, nitrogen, and sulfur. Which elements typically form cations? Some elements that typically form cations are generally metals such as Magnesium, Calcium, and, Barium. When an atom of Potassium ionizes, what charge will it become? When an atom of Potassium ionizes the charge becomes larger by one. Name at least two other elements whose atoms share the same charge when they ionize. Two Elements that share the same charge when they ionize are hydrogen and oxygen because when ionized they make water. Which family of elements gets a -1 charge when they ionize? The halogen family gets a -1 charge when it is ionized. Write the Calcium, Magnesium, Sodium, Chlorine, Oxygen, and Sulfur ions. Calcium +2, Magnesium +2, Sodium +1, Chlorine -1, Oxygen -2, and Sulfur -2 Name two ions that would combine in a 1:1 ratio. Two Ions that would combine to a 1:1 ratio would be Na and Cl (Sodium Chloride) Name two ions that would combine in a 2:1 ratio. Two elements would be Na2O (Sodium Oxide) Which family of elements gets a +1 charge when they ionize? Alkaline Earth Metals gets a +1 charge. Which family of elements gets a +2 charge when they ionize? Alkaline Earth Metals gets +2 charge. When coming across an article on CNN about Aliens it immediately got me hooked. I have always pondered the thought of life outside of earth. A part of me is Excited while the other half is scared out of my mind. Just the thought of coming into contact with an other form of life is scary to me. What if there trying to take over or what if there friendly. Ever since watching Star Wars it has opened my mind to these kind of thoughts. Maybe they can help our society advance faster than it has ever had before. After reading the article it has only made me believe more and more that maybe in my lifetime I could see them. For example the article states that there are over 40 billion planets out there that are Earth-Like meaning that there is a high possibility of life out there. As I kept reading the article it only made me believe more that soon we could talk to aliens. But It also made me realize that if do come into contact that they might be far more advanced than we are or maybe far behind us. Either way I find the concept of coming into contact with aliens interesting. Overall I found the article entertaining to read.
|



















 RSS Feed
RSS Feed